You use pharmaceutical software to speed development, reduce errors, and stay compliant across discovery, trials, and manufacturing. The right software centralizes data, automates repetitive tasks, and gives you reliable audit trails so you can move drugs from concept to clinic faster and with fewer regulatory risks.
This article breaks down the key features that matter—data management, LIMS, MES, analytics, and compliance tools—and shows where those capabilities fit across R&D, clinical trials, and production. Expect practical examples and forward-looking trends so you can evaluate solutions that match your workflow and scale with your programs.
Key Features of Pharmaceutical Software
You need software that enforces regulatory requirements, controls inventory and supply chains, records precise batch histories, and ensures product quality through automated checks and documentation. Each capability must map to specific processes you run daily, from raw-material receipt to finished-product release.
Regulatory Compliance Tools
Regulatory tools help you maintain electronic records and signatures that meet 21 CFR Part 11 and EU Annex 11 requirements. They include audit trails that record who changed what and when, version control for SOPs and documents, and configurable electronic signatures tied to user roles.
You should expect built-in validation support: validation scripts, test case libraries, and traceable IQ/OQ/PQ documentation templates that reduce vendor and internal testing time. Look for automated reporting of deviations, CAPA workflows, and regulatory submission exports (eCTD/XML) to streamline inspections and filings.
Integration with your quality management system (QMS) and LIMS prevents duplicate data entry. Role-based access, multifactor authentication, and data encryption protect records while enabling secure remote review during audits.
Inventory and Supply Chain Management
Inventory modules track material lots, expiry dates, and storage conditions across plants and warehouses. They support FIFO/LIFO allocation rules, automated reorder points, and safety stock calculations to help you avoid stockouts or expired material use.
Supplier qualification and audit records should live in the same system so you can tie incoming material lots to supplier certificates of analysis (CoAs). Look for ASN (Advanced Shipping Notice) and EDI integrations to automate receipt processing and reduce manual reconciliation.
Cold-chain management features monitor temperature excursions and generate alerts. Reporting dashboards should give you per-site days-on-hand, lot-level consumption rates, and traceability links between raw materials and finished goods for efficient recalls.
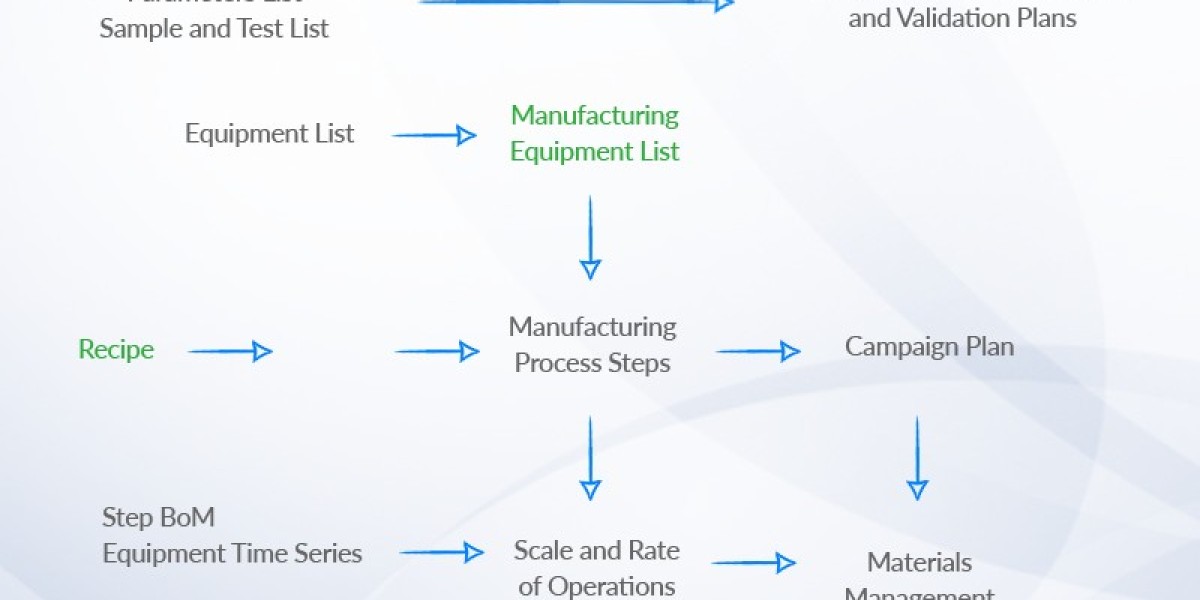
Batch Tracking and Traceability
Batch tracking provides a complete genealogy from raw material lot to finished product serial or batch number. You need lot-to-lot linkage, manufacturing step timestamps, operator IDs, and equipment IDs to recreate production history precisely.
Systems should support serialized tracking and GS1 standards (GTIN, GLN) for interoperability with distributors and regulators. Real-time lineage queries let you identify affected downstream batches fast during a recall or deviation investigation.
Ensure the software captures process parameters and critical control points automatically, either from shop-floor systems or manual input with validation rules. This data should be exportable in machine-readable formats for rapid analysis and regulatory response.
Quality Assurance Modules
QA modules automate release testing, deviation handling, CAPA, and change control workflows that you rely on to maintain compliance. They should link test results from your lab information system (LIMS) directly to batch records so you can conditionally release or quarantine product without delay.
Expect configurable quality gates and approval chains that enforce your policies, with deadline tracking and escalation rules to prevent backlog. Trend analysis and KPI widgets help you spot recurring issues — for example, OOS rates by supplier or equipment downtime correlating with defects.
Document control, training records, and supplier quality metrics should integrate with QA processes so you can demonstrate continuous improvement and meet inspection evidence requirements.
Applications and Future Trends
You will see software driving faster molecule discovery, smoother manufacturing, and stricter protection of sensitive data. Expect tools that automate repetitive work, surface actionable insights from large datasets, and enforce compliance across complex supply chains.
Drug Development and Clinical Trials
You can shorten discovery timelines by using AI models that predict binding affinity, generate candidate molecules, and prioritize leads for synthesis. Platforms that integrate high-throughput screening data, omics datasets, and historical trial outcomes let you triage compounds with quantitative confidence scores.
Clinical trial management systems now automate randomization, eConsent, and remote monitoring. You will capture wearable and ePRO (electronic patient‑reported outcomes) data in real time, reducing site visits while maintaining data integrity.
Adaptive trial designs and synthetic control arms rely on structured, interoperable data standards (CDISC, FHIR). You should require traceable audit trails, versioned datasets, and reproducible analysis pipelines to meet regulators’ expectations and speed approvals.
Production Automation
You can improve batch consistency and reduce downtime by deploying digital twins and closed‑loop control systems that link process models to live plant data. These systems perform model‑based adjustments to temperature, pH, and feed rates to keep critical quality attributes within limits.
Manufacturing execution systems (MES) and ERP integration let you trace materials from raw inputs to finished lots, enabling faster product recalls and regulatory reporting. Use predictive maintenance driven by sensor telemetry and anomaly detection to cut unplanned stoppages.
Implement role‑based access, electronic batch records, and automated deviation workflows so operators, QA, and supply planners work from a single source of truth and actions are recorded for inspection.
Data Security in Pharmaceuticals
You must protect intellectual property and patient data across development and commercial systems. Apply zero‑trust architecture, network segmentation, and multifactor authentication to limit lateral movement and unauthorized access.
Encrypt data at rest and in transit, and use hardware security modules (HSMs) or cloud key management for cryptographic keys. Maintain immutable audit logs and tamper‑evident storage for trial records and manufacturing data to satisfy inspectors.
Regularly run threat modeling, penetration testing, and supply‑chain assessments. Require vendor attestations for SaaS components and enforce secure CI/CD pipelines so code and models you deploy remain auditable and integrity‑protected.